Research Areas
Our lab specializes in neuromodulation technologies and neural circuit analysis for psychiatric disorders.
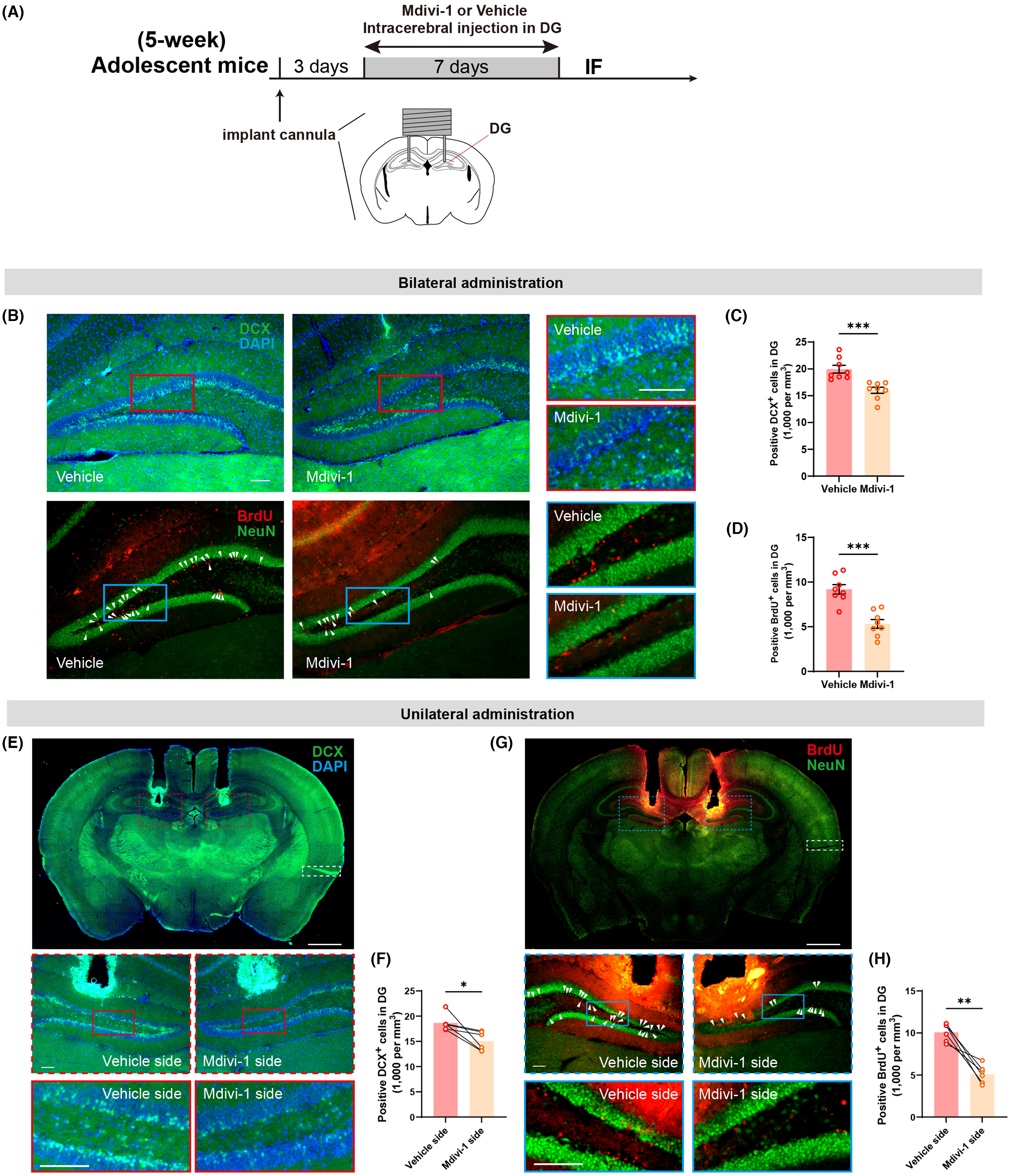
Neuromodulation Technologies
Our lab specializes in cutting-edge neuromodulation techniques for psychiatric disorders, integrating invasive and non-invasive approaches to modulate neural circuits and improve patient outcomes.
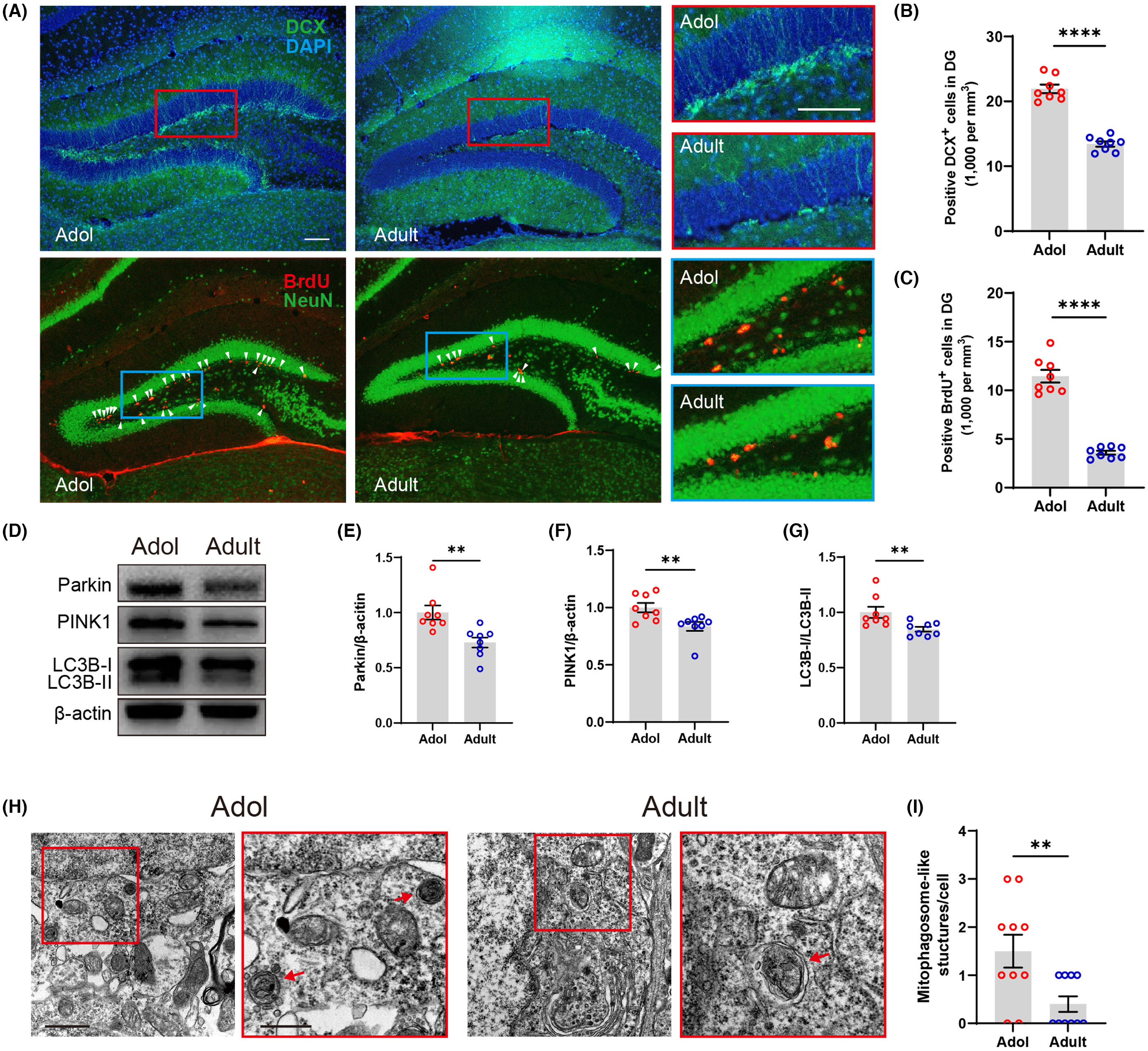
Deep Brain Stimulation (DBS): We investigate therapeutic mechanisms of DBS for treatment-resistant psychiatric disorders, translating research from rodent models to human applications using microelectrode arrays and electrophysiological recordings.
Patch Clamp & Electrophysiology: State-of-the-art patch-clamp techniques to study synaptic transmission, ion channel function, and neural excitability at the single-cell level.
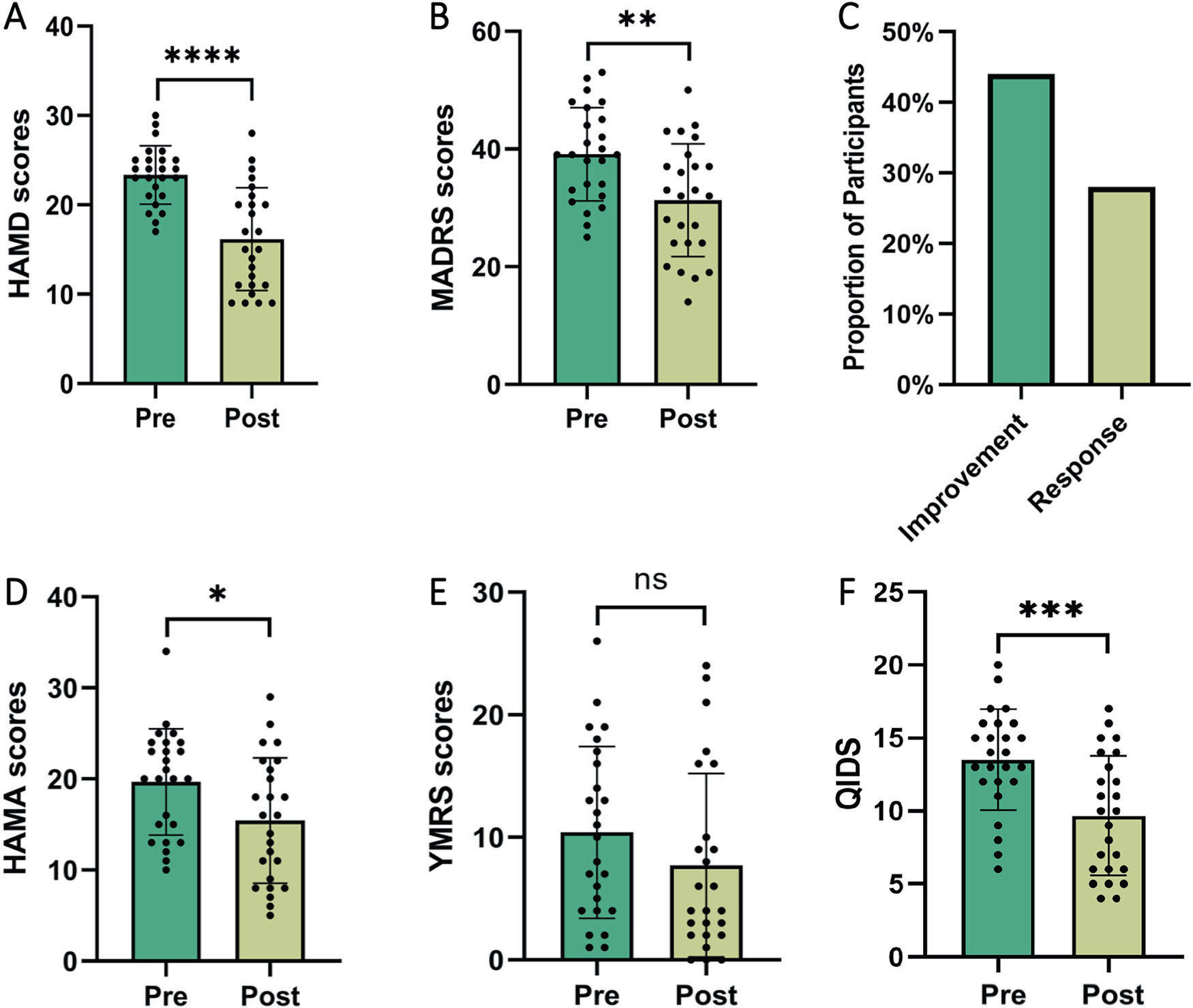
Repetitive Transcranial Magnetic Stimulation (rTMS): Development of personalized rTMS protocols with neuronavigation and functional connectivity targeting, demonstrated to significantly improve bipolar depression in clinical trials.
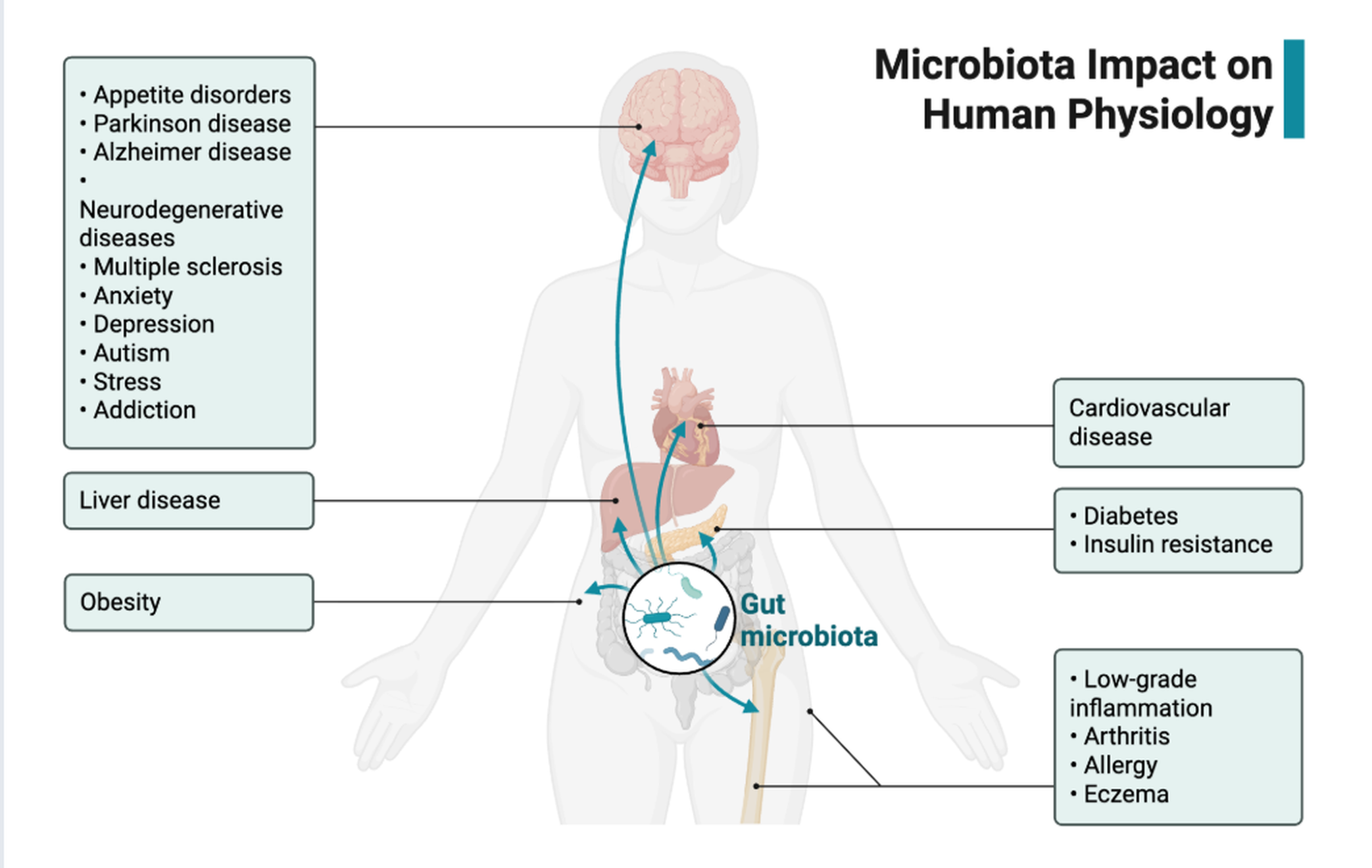
Microbiota-Gut-Brain Axis
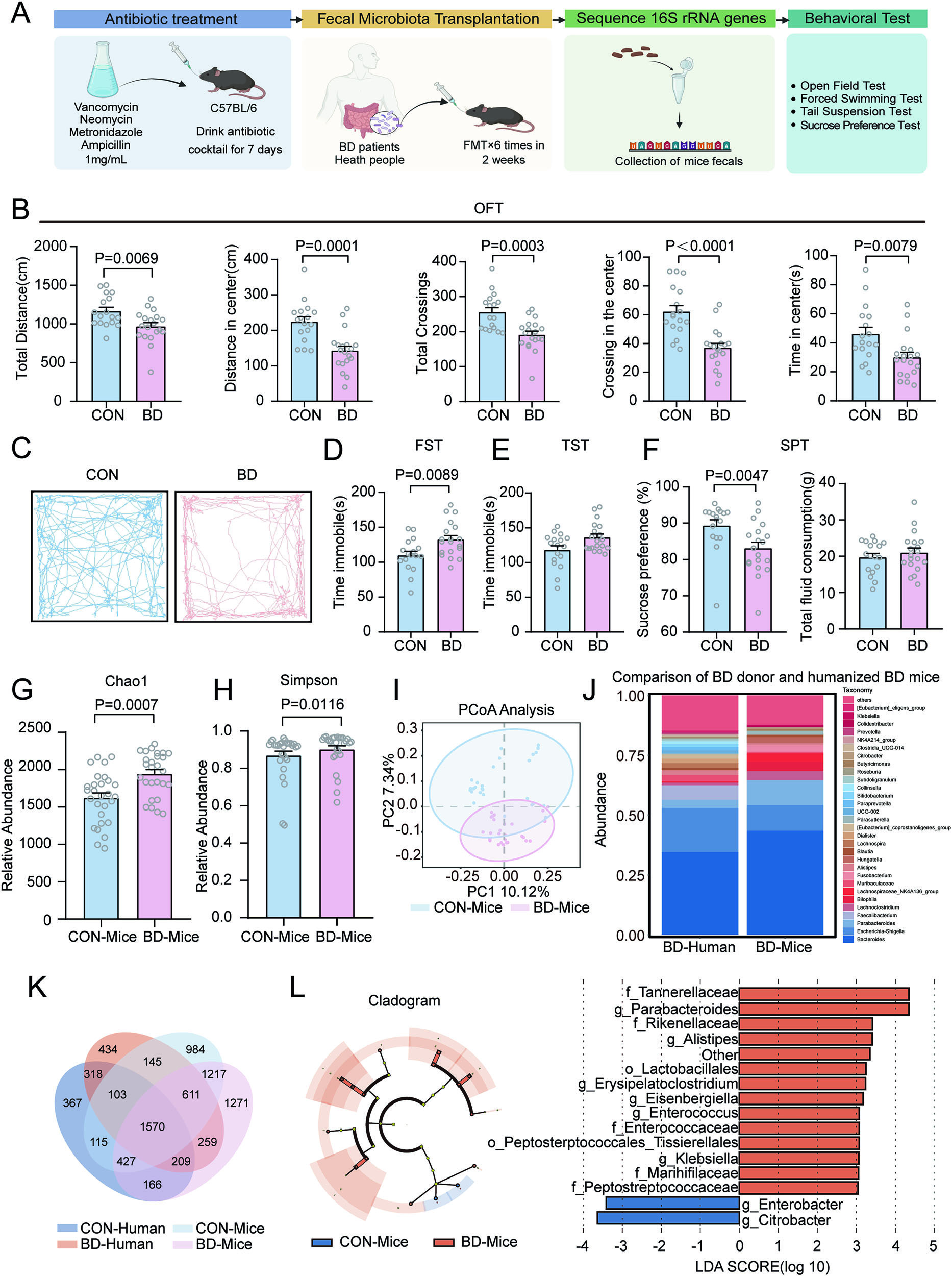
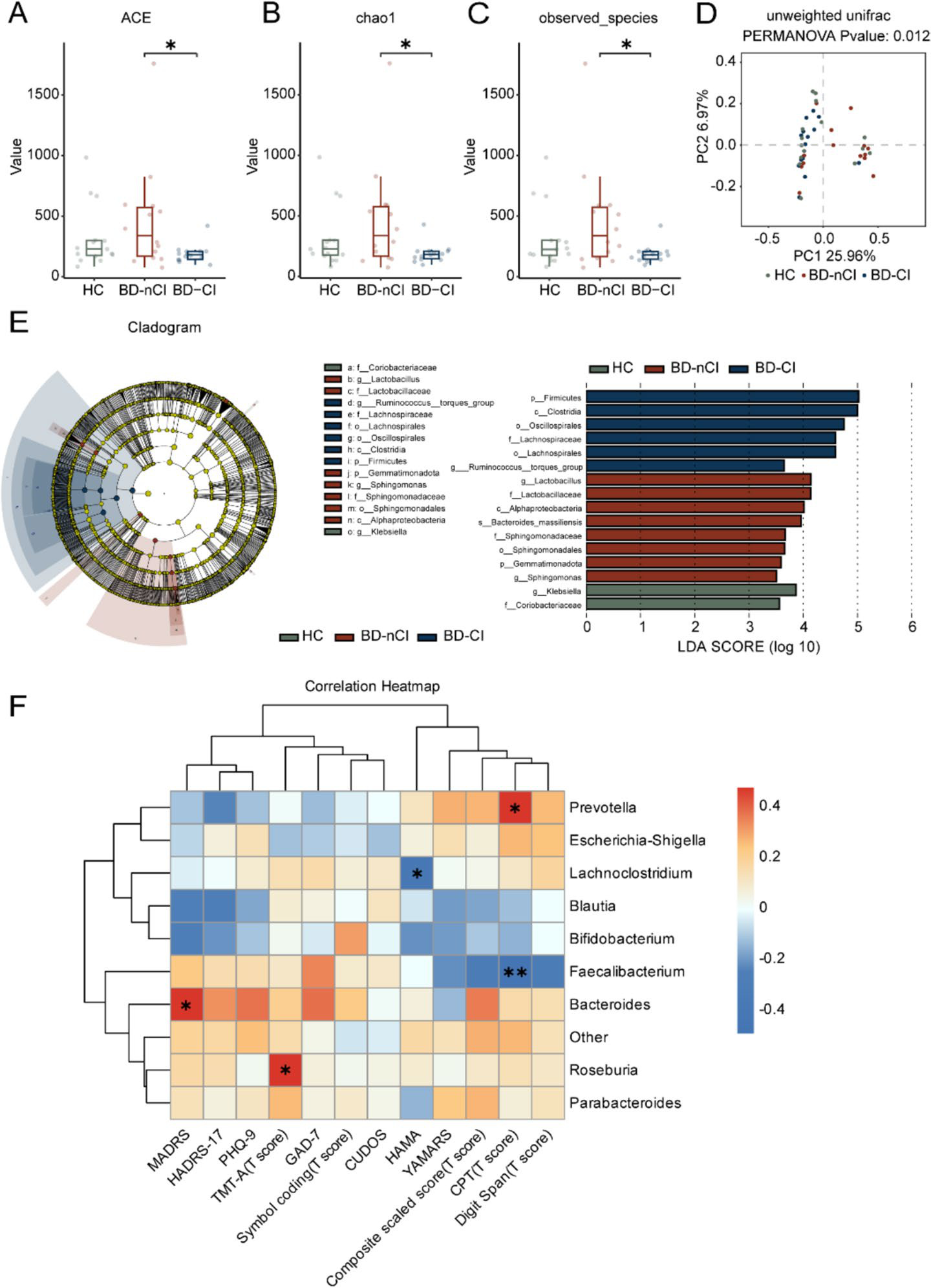
We explore the complex communication between gut microbiota and brain function in psychiatric disorders, with particular focus on bipolar depression and its underlying mechanisms.
Our research demonstrates that gut microbiota from bipolar depression patients can induce depression-like behaviors through modulation of synaptic connectivity and dopamine transmission in the VTA-mPFC pathway. We employ fecal microbiota transplantation, metagenomics, and metabolomics to uncover the causal role of gut microbes in mental health.